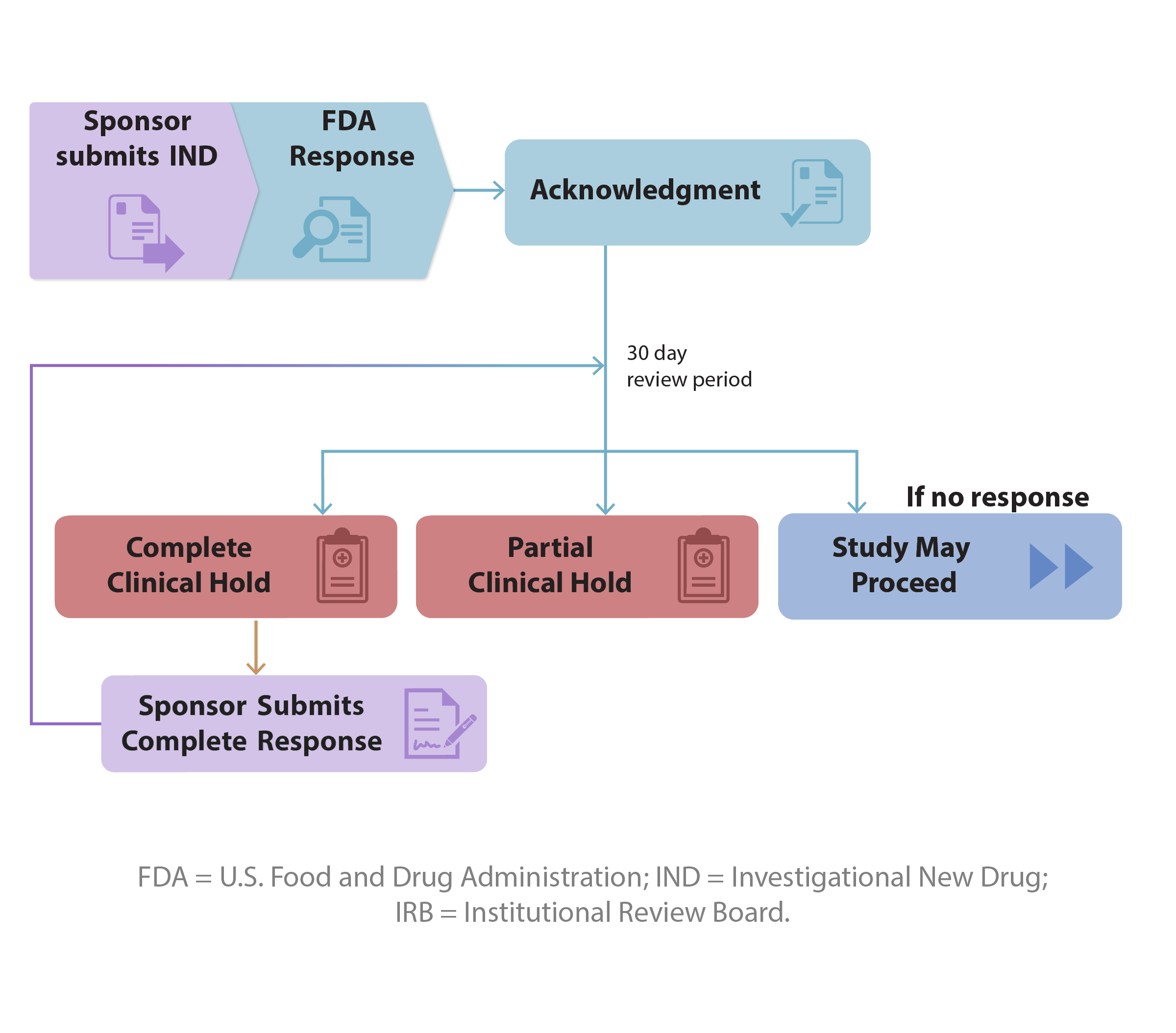
Upon receipt of an IND application, FDA will notify the sponsor of the date it receives the application through an IND acknowledgment letter. The acknowledgment letter will also contain the IND number, the FDA review division assigned to review the IND, and the FDA point of contact. The study may proceed 30 days after FDA receives the IND, unless FDA notifies the sponsor that the investigations described in the IND are subject to a clinical hold. In the absence of hearing from FDA, the sponsor is encouraged to contact FDA to ensure the study may proceed.
An order issued by FDA to the sponsor of an IND to delay or to suspend a clinical investigation. A clinical hold may be either a complete clinical hold or a partial clinical hold.
Complete Clinical Hold: A delay or suspension of all clinical work requested under an IND. If a sponsor submits an initial IND and within the first 30-day period FDA and the sponsor agree on an alternative protocol that is allowed to proceed, this does not constitute a clinical hold provided there are no specific FDA contingencies that require FDA review/approval before further studies are started.
Partial Clinical Hold: A delay or suspension of only part of the clinical work requested under the IND (e.g., a specific protocol or part of a protocol is not allowed to proceed; however, other protocols or parts of the protocol are allowed to proceed under the IND).
After an IND has been placed on clinical hold, the study may not be initiated or resume until the Agency has notified the sponsor (via phone, fax, letter, or e-mail) that the study may proceed (21 CFR 312.42(e)). (Communications by phone, fax, or e-mail will be followed by a letter.)
For more information regarding clinical holds and how to address them, please visit the FDA site , and the accompanying guidance .
The study may proceed 30 days after FDA receives the IND, unless FDA notifies the sponsor that the investigations described in the IND are subject to a clinical hold. In the absence of receiving a Study May Proceed letter from FDA, the sponsor is encouraged to contact FDA in writing to ensure the study may proceed.
Annual reports are due within 60 days of Study May Proceed anniversary date.