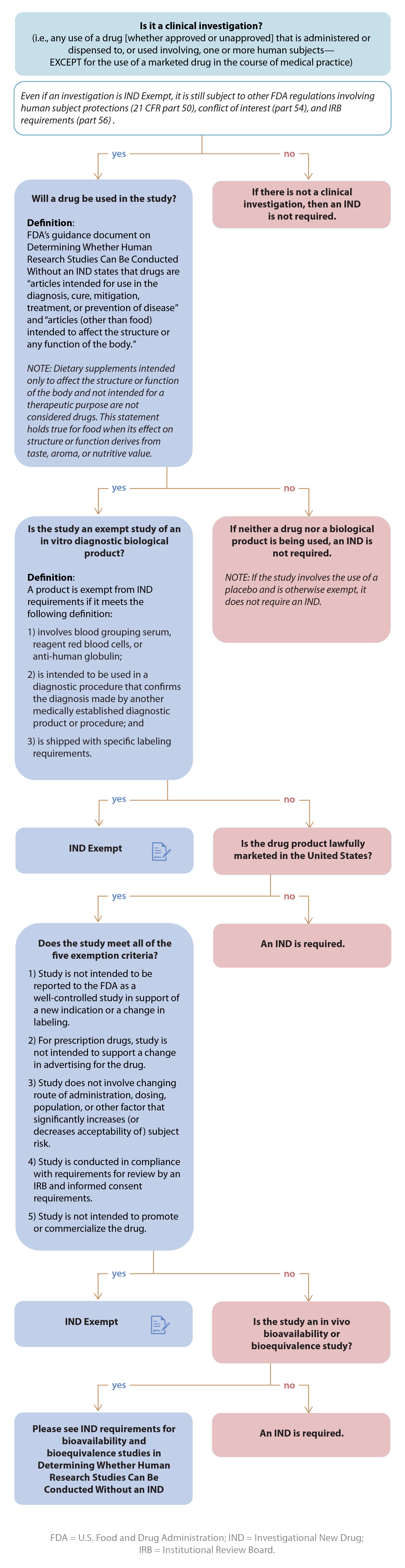
Is My Study Exempt?
What Is a Drug?
The definition of the term "drug" (section 201(g)(1) of the FD&C Act) includes:
"articles intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease . . ." and
"articles (other than food) intended to affect the structure or any function of the body of man or other animals."
Biological products are also considered drugs within the meaning of the FD&C Act. A biological product is:
"a virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or derivative, allergenic product, protein (except any chemically synthesized polypeptide), or analogous product, or arsphenamine or derivative of arsphenamine (or any other trivalent organic arsenic compound), applicable to the prevention, treatment, or cure of a disease or condition of human beings."
IND Exemption Criteria
Whether a clinical investigation of a marketed drug can be eligible for an IND exemption primarily depends on the intent of the investigation and the degree of risk associated with the use of the drug in the investigation. If the drug is not legally marketed, the study is not eligible for IND exemption and would require an IND.
For additional information please review The FDA Guidance: Investigational New Drug Applications (INDs) — Determining Whether Human Research Studies Can Be Conducted Without an IND .
A clinical investigation of a drug product that is lawfully marketed in the United States is exempt from the IND requirements if all of the criteria for an exemption in §312.2(b) are met:
The investigation does not involve a route of administration, dose, patient population, or other factor that significantly increases the risk (or decreases the acceptability of the risk) associated with the use of the drug product (21 CFR 312.2(b)(1)(iii)).
The investigation is not intended to be reported to FDA as a well-controlled study in support of a new indication and there is no intent to use it to support any other significant change in the labeling of the drug.
The investigation is conducted in compliance with the requirements of §312.7 (i.e., the investigation is not intended to promote or commercialize the drug product).
In the case of a prescription drug, the investigation is not intended to support a significant change in the advertising for the drug.
The investigation is conducted in compliance with the requirements for review by an IRB (21 CFR part 56) and with the requirements for informed consent (21 CFR part 50).
What is meant by a drug product that is lawfully marketed in the United States?
The product must be legally marketed as a drug, meaning it has a New Drug Application (NDA), Abbreviated New Drug Application (ANDA), or Biologics Licensing Application (BLA) that was approved by the FDA. The labeling or package insert details the approved indications for use and dosage and administration.
Most up-to-date drug labeling information
-
A study can be IND exempt even if the drug is not used in exactly the same dosage form, dosage levels, and patient populations described in the marketed labeling for the product. Changes to the lawfully marketed drug product that do not increase the risks over the risk presented by use of the product are allowed.
Sponsors or sponsor-investigators can make low-risk modifications to the lawfully marketed dosage form to, for example, blind a study.
Is the risk associated with the product significantly increased (or the acceptability of the risk significantly decreased)?
Investigators should carefully consider the risk implications of any conditions of use in the study that deviate from the conditions of use described in the drug's labeling, with particular attention to the following:
Route of Administration:
A change in the route of administration can introduce a significant new risk.
For example, there could be a significant increase in risk if a marketed drug for oral administration is converted to a dosage form that is to be administered by injection or intravenous, intrathecal, or inhalation route. These other routes of administration introduce concerns with increased local concentrations, sterility, pyrogenicity, hypersensitivity (e.g., airway reactivity), variations in metabolism, and other issues not present with oral administration that can significantly increase the risk, or decrease the acceptability of the risk, associated with use of the drug.
Dose:
Increases in dose, frequency, or duration of administration, compared to labeled dosing regimens, can significantly increase the risk in a study using a marketed drug.
It is also possible that a decrease in dose could significantly increase risk. For example, administering a sub-therapeutic dose of an antiviral drug to study subjects could induce resistance in the subjects, thus rendering a subsequent therapeutic dose of the drug ineffective in treating the virus.
The significance of changes in dose (in particular, increases in dose) can vary across therapeutic areas. For example, the cancer treatment guidance provides some latitude for conducting studies of high-dose cancer treatments without an IND because oncologists are generally familiar with the implications of high-dose regimens. In other clinical settings, use of higher doses than are recommended in labeling may be much more likely to significantly increase the risk or decrease the acceptability of the risk.
Patient Population:
The acceptability of known and unknown risks can vary across different treatment populations (see §312.2(b)(1)(iii)).
The population chosen for study could be at increased risk compared to the approved use population for a variety of reasons, such as increased age, different disease or stage of disease, concomitant illness, decreased renal or hepatic function, or concomitant therapy.
For example, a drug with significant toxicity can be approved for use in a population with a life-threatening or severely debilitating disease because the risk of toxicity is acceptable in that population.
Use of that drug in a clinical investigation in a population that is not so ill (e.g., to evaluate the drug for prevention of disease or symptomatic relief), however, would present a different risk-benefit situation in which the known risks might not be acceptable.
When use of the drug in a specific patient population decreases the acceptability of the known risks, the study would have to be conducted under an IND as required under 21 CFR part 312.
Does the sponsor intend to (1) report to FDA the investigation as a well-controlled study in support of a new indication, (2) use it to support any other significant change in the labeling of the drug, or (3) use it to support a significant change in the advertising (for prescription drugs only) for the drug?
Generally, any well-controlled trial of a marketed drug (e.g., a study of a new indication) sponsored by the manufacturer of the drug would be intended to be used to influence labeling or promotion in some significant way and would have to be conducted under an IND.
-
Studies of marketed drugs conducted by an entity that does not have an independent ability to change a drug's labeling – e.g., a study conducted by a sponsor-investigator in an academic setting or Government agency sponsor – would not generally be intended to be submitted to FDA to support a new indication or to otherwise influence the drug's labeling or promotion.
However, data from such studies may subsequently be submitted to FDA for that purpose and, therefore, FDA has an interest in helping to ensure that these studies are designed to yield data adequate to support a labeling change. A sponsor who would like to obtain FDA advice on study design can submit an IND for FDA review.
How to get an IND Exemption
An investigator who believes their study is eligible for an IND exemption can submit the study along with supportive rationale directly to the IRB. The IRB is able to grant an IND exemption directly.
If the IRB does not agree that the study is eligible for IND exemption, the FDA will need to make the final decision. Either a formal or an informal IND exemption request can be made to the FDA:
An informal IND exemption request by phone or email can be made to the reviewing division at FDA. However, it is up to the division if they are willing or able to provide feedback.
The formal process for requesting an IND exemption requires submitting a complete IND along with a request and rationale for the exemption in the cover letter.
Specific Issues Concerning the Application of the IND Regulations
Please review the FDA guidance for the following specific topics:
Dietary Supplements
Under the Dietary Supplement Health and Education Act of 1994 (DSHEA), a dietary supplement is defined, in part, as:
A product taken by mouth that is intended to supplement the diet and that contains one or more dietary ingredients.
The dietary ingredients in these products can include vitamins, minerals, herbs and other botanicals, amino acids, other dietary substances intended to supplement the diet, and concentrates, metabolites, constituents, extracts, or combinations of the preceding types of ingredients.
Dietary supplements can be found in many forms such as tablets, capsules, softgels, liquids, or powders.
Whether an IND is needed for a clinical investigation evaluating a dietary supplement is determined by the intent of the clinical investigation.
If the clinical investigation is intended only to evaluate the dietary supplement's effect on the structure or function of the body, an IND is not required.
For example: studying the effect of guarana on maximal oxygen uptake or the effect of fiber on bowel regularity
If the clinical investigation is intended to evaluate the dietary supplement's ability to diagnose, cure, mitigate, treat, or prevent a disease, an IND is required. For example: evaluating a supplement's ability to prevent osteoporosis or treat chronic diarrhea or constipation
Conventional Foods
For studies evaluating the effects of a conventional food, the need for an IND depends on the intent of the clinical investigation. As is the case for a dietary supplement, a food is considered to be a drug if it is "intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease," Therefore, a clinical investigation intended to evaluate the effect of a food on a disease requires an IND.
For example, a clinical investigation intended to evaluate the effect of a food on the signs and symptoms of Crohn's disease would require an IND.
Endogenous Compounds
A common question is whether provocation or challenge studies in which an endogenous compound (e.g., bradykinin, histamine, angiotensin) is administered to subjects to evoke a physiologic response, characterize a disease, or establish the mechanism of action are subject to IND requirements. In these cases, the endogenous compound is plainly not being used for a therapeutic purpose. There is, however, intent to affect the structure or function of the body, so the compound would be considered a drug under these circumstances. Therefore, these types of studies are clinical investigations and require an IND, unless the study meets the criteria for an exemption.
Live Organisms
An IND is required for challenge studies in which a live organism (e.g., virus, bacteria, or fungi, whether modified or wild-type) is administered to subjects to study the pathogenesis of disease or the host response to the organism. Although the challenge organism is not intended to have a therapeutic purpose, there is intent to affect the structure or function of the body. Thus, the organism is both a biological product (see 21 CFR 600.3(h)(1)) and a drug, and an IND is required for the clinical investigation, unless the criteria for exemption are met.
Cosmetics
Studies of ingredients or products marketed as cosmetics require an IND if the ingredient is being studied for use to affect the structure or function of the body or to prevent, treat, mitigate, cure, or diagnose a disease.
This is true even if the study is intended to support a cosmetic claim about the ingredient or product's ability to cleanse, beautify, promote attractiveness, or alter the appearance, rather than a structure/function claim.
For example, a study of the effect of a cosmetic product containing human or animal biological material (such as placenta) on skin repair mechanisms would require an IND, even if the study is intended only to support a claim of younger looking skin.
Bioavailability or Bioequivalence Studies
A BA/BE study in humans does not require an IND if all of the following conditions are met:
The drug product does not contain a new chemical entity (21 CFR 314.108), is not radioactively labeled, and is not cytotoxic.
The dose (single dose or total daily dose) does not exceed the dose specified in the labeling of the approved version of the drug product.
The investigation is conducted in compliance with the requirements for review by an IRB (21 CFR part 56) and with the requirements for informed consent (21 CFR part 50).
The sponsor meets the requirements for retention of test article samples (21 CFR 320.31(d)(1)) and safety reporting (21 CFR 320.31(d)(3)).
Radioactive Isotopes
Human research using a radioactive drug or biological product may be conducted without an IND if
It involves basic research not intended for immediate therapeutic, diagnostic, or similar purposes, or otherwise to determine the safety and efficacy of the product,
The use in humans is approved by a Radioactive Drug Research Committee (RDRC) that is composed and approved by FDA,
The dose to be administered is known not to cause any clinically detectable pharmacological effect in humans, and
The total amount of radiation to be administered as part of the study is the smallest radiation dose practical to perform the study without jeopardizing the benefits of the study and is within specified limits.
Cold Isotopes
FDA does not intend to object to clinical investigations using cold isotopes of unapproved drugs being conducted without an IND, provided the following conditions are met (the conditions are based on the criteria for studies using radiolabeled drugs (see 21 CFR 361.1))
The research is intended to obtain basic information regarding the metabolism (including kinetics, distribution, and localization) of a drug labeled with a cold isotope or regarding human physiology, pathophysiology, or biochemistry.
The research is not intended for immediate therapeutic, diagnostic, or preventive benefit to the study subject.
The dose to be administered is known not to cause any clinically detectable pharmacologic effect in humans based on clinical data from published literature or other valid human studies.
The quality of the cold isotope meets relevant quality standards.
The investigation is conducted in compliance with the requirements for review by an IRB (21 CFR part 56) and the requirements for informed consent (21 CFR part 50).
In Vitro Diagnostic Biological Products
A clinical investigation involving an in vitro diagnostic biological product (i.e., blood grouping serum, reagent red blood cells, or anti-human globulin) is exempt from IND requirements if (a) it is intended to be used in a diagnostic procedure that confirms the diagnosis made by another, medically established, diagnostic product or procedure and (b) it is shipped in compliance with 21CFR312.160.
Use of Placebos
A clinical investigation involving use of a placebo is exempt from IND requirements if the investigation does not otherwise require submission of an IND.